 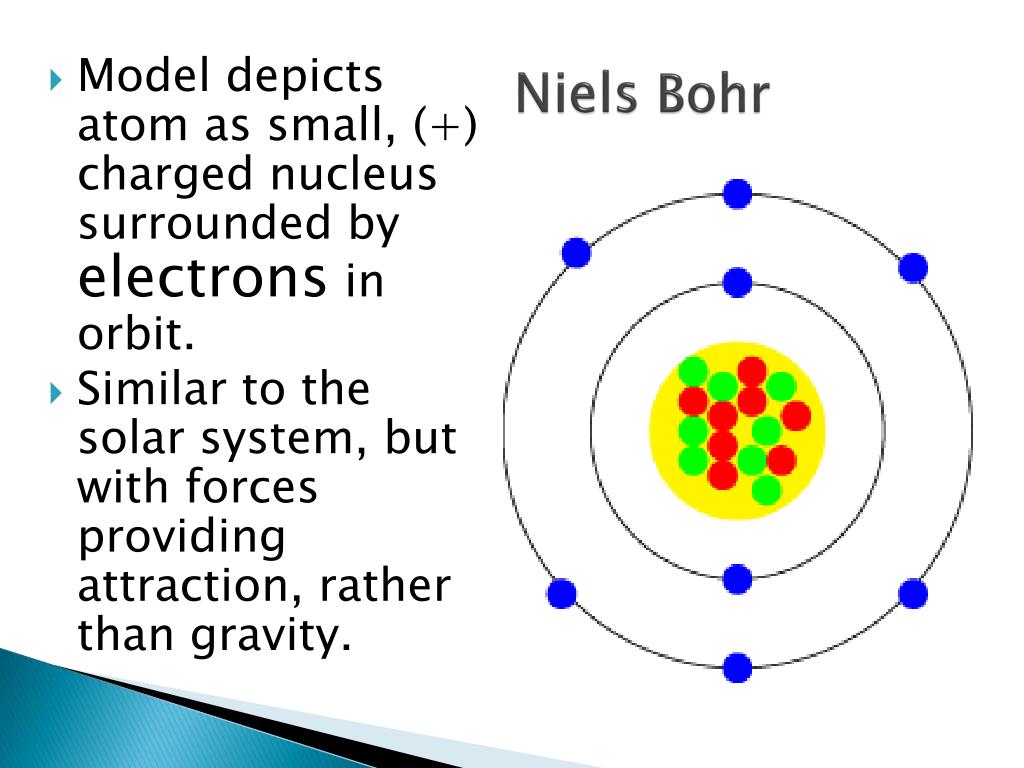
It features a 1-hour lecture video, and also presents the prerequisites, learning objectives, reading assignment, lecture slides, homework with solutions, and resources for further study. The Bohr model represents these energy levels as rings. This page contains materials for the session on the atomic models of Rutherford and Bohr. 
The shells are at certain distances from the nucleus. Bohr did calculations that led him to suggest that electrons orbit the nucleus in shells. Quantum theory revolutionized physics at the beginning of the 20th century, when Max Planck and Albert Einstein postulated that light energy is emitted or absorbed in discrete amounts known as quanta (singular, quantum). Sommerfeld argued that if electronic orbits. Einen ersten Überblick über die Atommodelle erhältst Du. Zu den bekanntesten Modellen zählen die Atommodelle nach Demokrit, Dalton, Thomson, Rutherford sowie Bohr und schließlich das heutige quantenmechanische Modell. Ein Atommodell ist der Versuch, sich den Aufbau von Atomen vorzustellen. BohrSommerfeld theory is named after Danish physicist Niels Bohr and German physicist Arnold Sommerfeld. Atommodelle Erklärung, Übersicht und Liste. This is an excellent idea, given the historic importance of Bohr’s 1913 work, which provided the bridge from Planck’s quantized interaction of matter and radiation (1900) to the full theory of quantum mechanics (1925-28). As well discuss later in the article, atomic electrons exist at specific energy levels. Niels Bohr adapted Ernest Rutherfords nuclear model. The BohrSommerfeld model (also known as the Sommerfeld model or BohrSommerfeld theory) was an extension of the Bohr model to allow elliptical orbits of electrons around an atomic nucleus. Many teachers like to introduce the Bohr atom toward the end of an introductory physics course. So, its easy to see that the atom above contains two electrons. \)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. The Bohr model represents the particle nature of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
